-
×
 Bed Pkg Se Innsp Fda Qr Pl End Lumex
1 × $1,094.00
Bed Pkg Se Innsp Fda Qr Pl End Lumex
1 × $1,094.00 -
×
 CranRx (R) Gummies
1 × $19.99
CranRx (R) Gummies
1 × $19.99 -
×
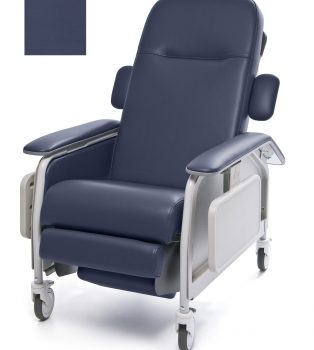 Recliner Cl Care Imperial Blue Ca133 New Ver Lumex
1 × $2,068.00
Recliner Cl Care Imperial Blue Ca133 New Ver Lumex
1 × $2,068.00 -
×
 #Sphyg Labstar Child (Gy) Latex Free, Labtron
1 × $20.00
#Sphyg Labstar Child (Gy) Latex Free, Labtron
1 × $20.00 -
×
 W/C Cushion Foam/Gel18X18X2.0 E & J Duragel Base 2
1 × $54.00
W/C Cushion Foam/Gel18X18X2.0 E & J Duragel Base 2
1 × $54.00 -
×
 Ice Cap English Style 6" Healthteam
1 × $7.00
Ice Cap English Style 6" Healthteam
1 × $7.00 -
×
 Alive! (R) energy Max water enhancer
1 × $8.99
Alive! (R) energy Max water enhancer
1 × $8.99 -
×
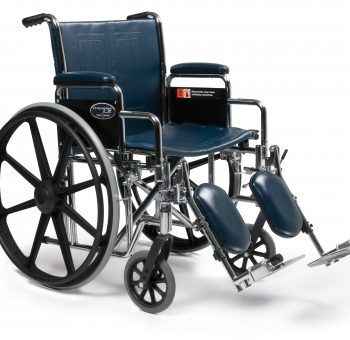 Traveler® Lx
1 × $420.00
Traveler® Lx
1 × $420.00 -
×
 Bath Seat W/Back Sea Gray - Lumex Unassembled
1 × $65.50
Bath Seat W/Back Sea Gray - Lumex Unassembled
1 × $65.50 -
×
 W/C Cushion Foam/Gel18X16X3.0 E & J Duragel Base 3
1 × $60.00
W/C Cushion Foam/Gel18X16X3.0 E & J Duragel Base 3
1 × $60.00 -
×
 W/C Trav Se 16X16 Fxd D-Ftr E&J
1 × $248.00
W/C Trav Se 16X16 Fxd D-Ftr E&J
1 × $248.00 -
×
 W/C Cushion Foam/Gel 20X16X2.0 E & J Duragel Base 2
1 × $54.00
W/C Cushion Foam/Gel 20X16X2.0 E & J Duragel Base 2
1 × $54.00 -
×
 W/C Cushion Foam/Gel20X18X2.0 E & J Duragel Base 2
1 × $56.00
W/C Cushion Foam/Gel20X18X2.0 E & J Duragel Base 2
1 × $56.00 -
×
 W/C Trav Se 18X16 Full Elr E&J
1 × $278.00
W/C Trav Se 18X16 Full Elr E&J
1 × $278.00 -
×
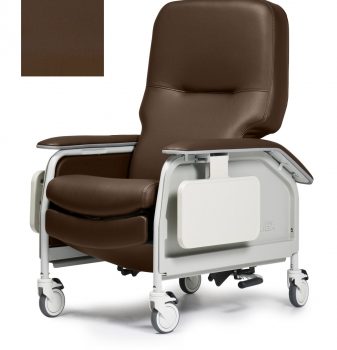 Recliner Dlx Cl Care Chestnut Ca133 Lumex
1 × $2,290.00
Recliner Dlx Cl Care Chestnut Ca133 Lumex
1 × $2,290.00 -
×
 W/C Cushion Gel/Foam16X16X4.5 E & J Duragel Spp
1 × $124.00
W/C Cushion Gel/Foam16X16X4.5 E & J Duragel Spp
1 × $124.00 -
×
 W/C Cushngeneral Use 22X18X3 E & J Duragel Base 3
1 × $64.00
W/C Cushngeneral Use 22X18X3 E & J Duragel Base 3
1 × $64.00 -
×
 W/C Trav Se 20X16 Full Elr E&J
1 × $288.00
W/C Trav Se 20X16 Full Elr E&J
1 × $288.00 -
×
 Ginkgold (R)
1 × $40.49
Ginkgold (R)
1 × $40.49 -
×
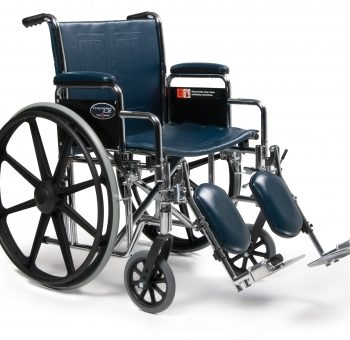 W/C Trav Lx 18X16 Dsk Elr E&J
1 × $370.00
W/C Trav Lx 18X16 Dsk Elr E&J
1 × $370.00 -
×
 B&T Triflora (R) Arthritis Gel††
1 × $7.49
B&T Triflora (R) Arthritis Gel††
1 × $7.49 -
×
 Lumbar Cushion (Foam) 22X19 Lumex
1 × $68.00
Lumbar Cushion (Foam) 22X19 Lumex
1 × $68.00 -
×
 W/C Cushion Foa/Foam 20X18X3.0 E & J Duragel Base 3
1 × $64.00
W/C Cushion Foa/Foam 20X18X3.0 E & J Duragel Base 3
1 × $64.00 -
×
 Recliner Drop Arm Eucalyptus Ca133 Lumex
1 × $2,388.00
Recliner Drop Arm Eucalyptus Ca133 Lumex
1 × $2,388.00 -
×
 W/C Cushion Foam/Gel18X16X2.0 E & J Duragel Base 2
1 × $52.00
W/C Cushion Foam/Gel18X16X2.0 E & J Duragel Base 2
1 × $52.00 -
×
 W/C Trav Se 20X16 Full D-Ftr E&J
1 × $268.00
W/C Trav Se 20X16 Full D-Ftr E&J
1 × $268.00 -
×
 W/C Trav Se 20X16 Dsk Elr E&J
1 × $288.00
W/C Trav Se 20X16 Dsk Elr E&J
1 × $288.00 -
×
 Recliner Drop Arm Periwinkle Ca133 Lumex
1 × $2,388.00
Recliner Drop Arm Periwinkle Ca133 Lumex
1 × $2,388.00 -
×
 W/C Trav Se 16X16 Dsk Elr E&J
1 × $278.00
W/C Trav Se 16X16 Dsk Elr E&J
1 × $278.00 -
×
 W/C Trav Se 16X16 Dsk D-Ftr E&J
1 × $258.00
W/C Trav Se 16X16 Dsk D-Ftr E&J
1 × $258.00 -
×
 B-100 Complex
1 × $13.99
B-100 Complex
1 × $13.99 -
×
 B-50 Complex
1 × $9.49
B-50 Complex
1 × $9.49 -
×
 Barley Grass Bulk Powder
1 × $26.99
Barley Grass Bulk Powder
1 × $26.99 -
×
 B&T SciaticAide (TM)
1 × $11.49
B&T SciaticAide (TM)
1 × $11.49 -
×
 B&T Daytime Cough & Bronchial Syrup
1 × $8.49
B&T Daytime Cough & Bronchial Syrup
1 × $8.49 -
×
 B&T Daytime Cough & Bronchial Syrup
1 × $13.99
B&T Daytime Cough & Bronchial Syrup
1 × $13.99 -
×
 B&T Nighttime Cough & Bronchial Syrup
1 × $8.49
B&T Nighttime Cough & Bronchial Syrup
1 × $8.49 -
×
 Vitamin E 400 IU
1 × $14.49
Vitamin E 400 IU
1 × $14.49 -
×
 Nettle Leaf
1 × $9.99
Nettle Leaf
1 × $9.99 -
×
 Vitamin C 500 mg with Bioflavonoids
1 × $10.49
Vitamin C 500 mg with Bioflavonoids
1 × $10.49 -
×
 MEGA 3/6/9 Omega Blend
1 × $41.49
MEGA 3/6/9 Omega Blend
1 × $41.49 -
×
 MSM 1000 mg
1 × $16.99
MSM 1000 mg
1 × $16.99 -
×
 Niacin
1 × $5.99
Niacin
1 × $5.99 -
×
 Niacinamide
1 × $7.99
Niacinamide
1 × $7.99 -
×
 MSM 1000 mg
1 × $20.99
MSM 1000 mg
1 × $20.99 -
×
 Melissa Lemon Balm Leaf
1 × $9.49
Melissa Lemon Balm Leaf
1 × $9.49 -
×
 NatureWorks (R) Swedish Bitters
1 × $40.99
NatureWorks (R) Swedish Bitters
1 × $40.99 -
×
 White Oak Bark
1 × $9.49
White Oak Bark
1 × $9.49 -
×
 Masquelier's (R) Tru-OPCs (TM)
1 × $21.49
Masquelier's (R) Tru-OPCs (TM)
1 × $21.49 -
×
 Vitamin D3 2,000 IU
2 × $8.99
Vitamin D3 2,000 IU
2 × $8.99 -
×
 Yucca Stalk
1 × $15.49
Yucca Stalk
1 × $15.49 -
×
 Wild Yam Root
1 × $9.99
Wild Yam Root
1 × $9.99
Subtotal: $14,035.73

