-
×
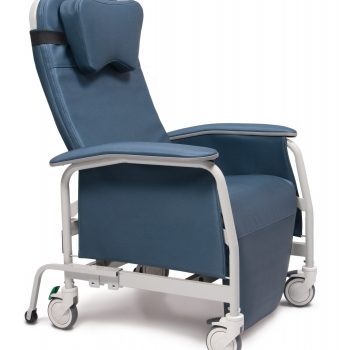 Recliner Pc Xwide Gypsum Ca-133, Lumex
1 × $1,776.00
Recliner Pc Xwide Gypsum Ca-133, Lumex
1 × $1,776.00 -
×
 #Aneroid Sphyg W/Thigh Cuf Lot
1 × $27.38
#Aneroid Sphyg W/Thigh Cuf Lot
1 × $27.38 -
×
 Trach Jcksn Impv Stnls Stl #6 John Bunn
1 × $118.66
Trach Jcksn Impv Stnls Stl #6 John Bunn
1 × $118.66 -
×
 Recl Dlx Heat&Mas Vintage Gold Ca-133, Lumex
1 × $3,474.00
Recl Dlx Heat&Mas Vintage Gold Ca-133, Lumex
1 × $3,474.00 -
×
 W/C Tr L4 18X16 Adj Dsk Elr Qr E&J
1 × $440.00
W/C Tr L4 18X16 Adj Dsk Elr Qr E&J
1 × $440.00 -
×
 Alive! (R) Max Potency Capsules
1 × $15.99
Alive! (R) Max Potency Capsules
1 × $15.99 -
×
 Recliner Pc Inf Pos Rose Ca133 Lumex
1 × $998.00
Recliner Pc Inf Pos Rose Ca133 Lumex
1 × $998.00 -
×
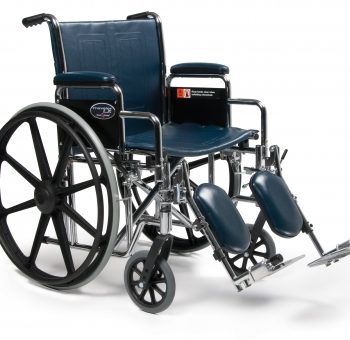 W/C Trav Lx 18X16 Full D-Ftr E&J
1 × $360.00
W/C Trav Lx 18X16 Full D-Ftr E&J
1 × $360.00 -
×
 Hydraplenish (TM) Hyaluronic Acid Serum
1 × $41.99
Hydraplenish (TM) Hyaluronic Acid Serum
1 × $41.99 -
×
 Traveler ®
1 × $471.00
Traveler ®
1 × $471.00 -
×
 Analog Tens With Timer Grafco
1 × $65.00
Analog Tens With Timer Grafco
1 × $65.00 -
×
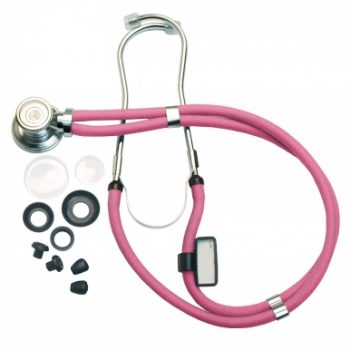 Steth Sprague-Rap 22" Neon Pk Labtron
1 × $14.38
Steth Sprague-Rap 22" Neon Pk Labtron
1 × $14.38 -
×
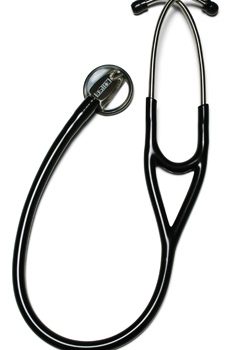 #Steth, 2 Tone Cardiol Blk Labtron
1 × $45.76
#Steth, 2 Tone Cardiol Blk Labtron
1 × $45.76 -
×
 #Steth Sprague-Rap 22" Lav Labtron
1 × $18.44
#Steth Sprague-Rap 22" Lav Labtron
1 × $18.44
Subtotal: $7,866.60

