-
×
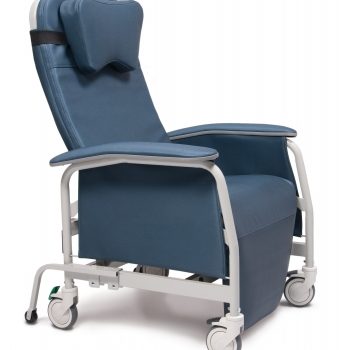 Recliner Pc Xwide Vintage Gold Ca-133, Lumex
1 × $1,776.00
Recliner Pc Xwide Vintage Gold Ca-133, Lumex
1 × $1,776.00 -
×
 Vitex Fruit
1 × $8.49
Vitex Fruit
1 × $8.49 -
×
 Willow Standardized
1 × $23.99
Willow Standardized
1 × $23.99 -
×
 Vitamin C 500 mg with Bioflavonoids
1 × $10.49
Vitamin C 500 mg with Bioflavonoids
1 × $10.49 -
×
 Vitamin C-500 with Rose Hips
2 × $9.49
Vitamin C-500 with Rose Hips
2 × $9.49 -
×
 Wild Yam Root
1 × $16.49
Wild Yam Root
1 × $16.49 -
×
 Yucca Stalk
2 × $10.49
Yucca Stalk
2 × $10.49 -
×
 Fracture Kit Grafco
1 × $189.48
Fracture Kit Grafco
1 × $189.48 -
×
 Vitamin B2
1 × $8.49
Vitamin B2
1 × $8.49 -
×
 Vitamin D3 5,000 IU
1 × $18.99
Vitamin D3 5,000 IU
1 × $18.99 -
×
 Zinc Lozenges
3 × $5.99
Zinc Lozenges
3 × $5.99 -
×
 Vitamin C-500 with Rose Hips
1 × $20.99
Vitamin C-500 with Rose Hips
1 × $20.99 -
×
 Willow
3 × $9.99
Willow
3 × $9.99 -
×
 White Oak Bark
2 × $9.49
White Oak Bark
2 × $9.49 -
×
 Yucca Stalk
2 × $15.49
Yucca Stalk
2 × $15.49 -
×
 W/C Rsc Ss Hi-Bk Pin Lk E&J
1 × $1,382.00
W/C Rsc Ss Hi-Bk Pin Lk E&J
1 × $1,382.00 -
×
 Chair Pressure Pad For P Alarm Lumex
2 × $47.00
Chair Pressure Pad For P Alarm Lumex
2 × $47.00 -
×
 Vitamin D3 2,000 IU
2 × $8.99
Vitamin D3 2,000 IU
2 × $8.99 -
×
 Wild Yam Root
1 × $9.99
Wild Yam Root
1 × $9.99 -
×
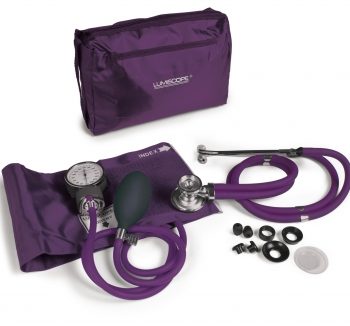 Spec Match Bp Cuff & Sprague Grape Lot #
1 × $32.16
Spec Match Bp Cuff & Sprague Grape Lot #
1 × $32.16 -
×
 Vitamin D3, 400 IU
1 × $8.49
Vitamin D3, 400 IU
1 × $8.49 -
×
 Head Halter Universal Grafco
1 × $10.48
Head Halter Universal Grafco
1 × $10.48 -
×
 Patient Alrm Advncd W/ Chr Pad Lumex
1 × $163.50
Patient Alrm Advncd W/ Chr Pad Lumex
1 × $163.50 -
×
 Trapeze Bariatric 600Lb Wt
1 × $1,080.00
Trapeze Bariatric 600Lb Wt
1 × $1,080.00
Subtotal: $5,009.87

