-
×
 Wheels 5 Swvl For 1 Wkrs Lumex
1 × $52.00
Wheels 5 Swvl For 1 Wkrs Lumex
1 × $52.00 -
×
 Recliner Pc Drp Arm Rosewood Ca133 Lumex
1 × $999.00
Recliner Pc Drp Arm Rosewood Ca133 Lumex
1 × $999.00 -
×
 Forearm Crutch Small Lumex
1 × $56.00
Forearm Crutch Small Lumex
1 × $56.00 -
×
 Iv Pole Bed Socket Tele 1 Ea Lumex
1 × $77.40
Iv Pole Bed Socket Tele 1 Ea Lumex
1 × $77.40 -
×
 Standard Glide Brakes Lumex
1 × $27.50
Standard Glide Brakes Lumex
1 × $27.50 -
×
 Bed Pkg Fe Rol Foam Mat Fda Qr Lumex
1 × $1,264.00
Bed Pkg Fe Rol Foam Mat Fda Qr Lumex
1 × $1,264.00 -
×
 Forearm/Platform Walker Attach Lumex
2 × $135.50
Forearm/Platform Walker Attach Lumex
2 × $135.50 -
×
 Crutch Forearm Lrg Ortho-Ease Lumex
1 × $68.00
Crutch Forearm Lrg Ortho-Ease Lumex
1 × $68.00 -
×
 W/C Trav L4 16X16 Dsk D-Ftr E&J
1 × $350.00
W/C Trav L4 16X16 Dsk D-Ftr E&J
1 × $350.00 -
×
 W/C Tr L4 16X16 Adj Dsk Ftr Qr E&J
1 × $420.00
W/C Tr L4 16X16 Adj Dsk Ftr Qr E&J
1 × $420.00 -
×
 Aneroid Sphyg Lot#
1 × $20.46
Aneroid Sphyg Lot#
1 × $20.46 -
×
 W/C Tr L4 16X16 Adj Dsk Elr Qr E&J
1 × $440.00
W/C Tr L4 16X16 Adj Dsk Elr Qr E&J
1 × $440.00 -
×
 Lumbar Cushion (Foam) 16X17 Lumex
1 × $52.00
Lumbar Cushion (Foam) 16X17 Lumex
1 × $52.00 -
×
 3" Swivel Wheels Lumex
1 × $38.00
3" Swivel Wheels Lumex
1 × $38.00 -
×
 Elevating Legrest With Composite Footplate
1 × $137.50
Elevating Legrest With Composite Footplate
1 × $137.50 -
×
 W/C Tr L4 18X16 Adj Dsk Elr Qr E&J
1 × $440.00
W/C Tr L4 18X16 Adj Dsk Elr Qr E&J
1 × $440.00 -
×
 W/C Trav L4 16X16 Dsk Elr E&J
1 × $370.00
W/C Trav L4 16X16 Dsk Elr E&J
1 × $370.00 -
×
 W/C Paramount Xd 26W Dsk Elr E&J
1 × $1,300.00
W/C Paramount Xd 26W Dsk Elr E&J
1 × $1,300.00 -
×
 W/C Tr L4 18X16 Adj Dsk Ftr Qr E&J
1 × $420.00
W/C Tr L4 18X16 Adj Dsk Ftr Qr E&J
1 × $420.00 -
×
 W/C Advant Rcl 18X17 Dsk Elr Notify Cust Of High Freight
1 × $530.00
W/C Advant Rcl 18X17 Dsk Elr Notify Cust Of High Freight
1 × $530.00 -
×
 W/C Advant Rcl 16X17 Dsk Elr Notify Cust Of High Freight
1 × $530.00
W/C Advant Rcl 16X17 Dsk Elr Notify Cust Of High Freight
1 × $530.00 -
×
 W/C Paramount Xd 30W Dsk Elr E&J
1 × $1,620.00
W/C Paramount Xd 30W Dsk Elr E&J
1 × $1,620.00 -
×
 W/C Trav L4 18X16 Full D-Ftr E&J
1 × $350.00
W/C Trav L4 18X16 Full D-Ftr E&J
1 × $350.00 -
×
 W/C Advant Rcl 18X17 Full Elr Notify Cust Of High Freight
1 × $530.00
W/C Advant Rcl 18X17 Full Elr Notify Cust Of High Freight
1 × $530.00 -
×
 W/C Paramount Xd 30W Dsk Swf E&J
1 × $1,510.00
W/C Paramount Xd 30W Dsk Swf E&J
1 × $1,510.00 -
×
 W/C Trav L4 18X16 Full Elr E&J
1 × $370.00
W/C Trav L4 18X16 Full Elr E&J
1 × $370.00 -
×
 W/C Advant Rcl 16X17 Full Elr Notify Cust Of High Freight
1 × $530.00
W/C Advant Rcl 16X17 Full Elr Notify Cust Of High Freight
1 × $530.00 -
×
 W/C Trav L4 20X16 Full D-Ftr E&J
1 × $360.00
W/C Trav L4 20X16 Full D-Ftr E&J
1 × $360.00 -
×
 W/C Travlr Hd 20X18 Full D-Ftr E&J
1 × $460.00
W/C Travlr Hd 20X18 Full D-Ftr E&J
1 × $460.00 -
×
 W/C Advantage Vnl18X16 Fxd Ftr Vinyl E&J
1 × $220.00
W/C Advantage Vnl18X16 Fxd Ftr Vinyl E&J
1 × $220.00 -
×
 W/C Trav L4 18X18 Full Elr E&J
1 × $374.00
W/C Trav L4 18X18 Full Elr E&J
1 × $374.00 -
×
 W/C Paramount Xd 26W Dsk Swf E&J
1 × $1,180.00
W/C Paramount Xd 26W Dsk Swf E&J
1 × $1,180.00 -
×
 W/C Trav Hd 22X18 Dsk D-Ftr E&J
1 × $470.00
W/C Trav Hd 22X18 Dsk D-Ftr E&J
1 × $470.00 -
×
 W/C Trans Bariatric Chair 24W Al E&J **On Temporary Hold**
1 × $440.00
W/C Trans Bariatric Chair 24W Al E&J **On Temporary Hold**
1 × $440.00 -
×
 W/C Trav L4 20X18 Dsk D-Ftr E&J
1 × $364.00
W/C Trav L4 20X18 Dsk D-Ftr E&J
1 × $364.00 -
×
 W/C Trav Hd 20X18 Full Elr E&J
1 × $480.00
W/C Trav Hd 20X18 Full Elr E&J
1 × $480.00 -
×
 W/C Trav L4 18X18 Dsk Elr E&J
1 × $374.00
W/C Trav L4 18X18 Dsk Elr E&J
1 × $374.00 -
×
 W/C Advantage 20X16 Dsk Elr E&J - Nylon Upholstery
1 × $256.00
W/C Advantage 20X16 Dsk Elr E&J - Nylon Upholstery
1 × $256.00 -
×
 W/C Htc 22X18 Desk Elr E&J
1 × $550.00
W/C Htc 22X18 Desk Elr E&J
1 × $550.00 -
×
 W/C Tr L4 18X18 Adj Dsk Ftr Qr E&J
1 × $424.00
W/C Tr L4 18X18 Adj Dsk Ftr Qr E&J
1 × $424.00 -
×
 W/C Trans Chair Green Alum 17" E&J
1 × $228.00
W/C Trans Chair Green Alum 17" E&J
1 × $228.00 -
×
 W/C Trav Hd 20X18 Dsk D-Ftr E&J
1 × $460.00
W/C Trav Hd 20X18 Dsk D-Ftr E&J
1 × $460.00 -
×
 W/C Advantage 16X16 Full D-Ftr E&J - Nylon Upholstery
1 × $226.00
W/C Advantage 16X16 Full D-Ftr E&J - Nylon Upholstery
1 × $226.00 -
×
 W/C Advantage 16X16 Dsk Elr E&J - Nylon Upholstery
1 × $246.00
W/C Advantage 16X16 Dsk Elr E&J - Nylon Upholstery
1 × $246.00 -
×
 W/C Advantage Vnl18X16 Fxd Elr Vinyl E&J
1 × $240.00
W/C Advantage Vnl18X16 Fxd Elr Vinyl E&J
1 × $240.00 -
×
 W/C Advantage 16X16 Fxd Elr E&J - Nylon Upholstery
1 × $236.00
W/C Advantage 16X16 Fxd Elr E&J - Nylon Upholstery
1 × $236.00 -
×
 Comfort Cushion 20X18X3 E&J
1 × $34.00
Comfort Cushion 20X18X3 E&J
1 × $34.00 -
×
 W/C Cushion Foam/Gel 20X16X3.0 E & J Duragel Base 3
1 × $62.00
W/C Cushion Foam/Gel 20X16X3.0 E & J Duragel Base 3
1 × $62.00 -
×
 W/C Cushion Gel/Foam 20X16X4.5 E & J Duragel Spp
1 × $134.00
W/C Cushion Gel/Foam 20X16X4.5 E & J Duragel Spp
1 × $134.00 -
×
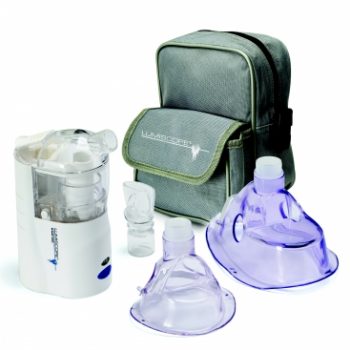 Portable Ultrasonic Nebulizer
1 × $19.95
Portable Ultrasonic Nebulizer
1 × $19.95 -
×
 Neb-U-Tyke Ic - Penguin Pediatric Nebulizer
1 × $57.00
Neb-U-Tyke Ic - Penguin Pediatric Nebulizer
1 × $57.00 -
×
 Neb-U-Tyke Bella Butterfly John Bunn
1 × $61.90
Neb-U-Tyke Bella Butterfly John Bunn
1 × $61.90 -
×
 Floor Stnd 2800A Chrm Pltd Lumex
1 × $142.00
Floor Stnd 2800A Chrm Pltd Lumex
1 × $142.00 -
×
 Trapeze Bariatric 450 Lb Wt
1 × $720.00
Trapeze Bariatric 450 Lb Wt
1 × $720.00 -
×
 Patient Alarm Basic W/ Bed Pad Lumex
1 × $163.50
Patient Alarm Basic W/ Bed Pad Lumex
1 × $163.50 -
×
 Yarrow Flowers
2 × $5.99
Yarrow Flowers
2 × $5.99 -
×
 Cart 2 Tier Versacart, 5" Cstr Lumex
1 × $227.52
Cart 2 Tier Versacart, 5" Cstr Lumex
1 × $227.52 -
×
 Vitamin E 400 IU
1 × $14.49
Vitamin E 400 IU
1 × $14.49
Subtotal: $22,009.20

