-
×
 Bed Pkg Se Innerspring Fda Fr Lumex
1 × $1,060.00
Bed Pkg Se Innerspring Fda Fr Lumex
1 × $1,060.00 -
×
 Bed Pkg Se Roll Foam Fr Pl End Lumex
1 × $1,134.00
Bed Pkg Se Roll Foam Fr Pl End Lumex
1 × $1,134.00 -
×
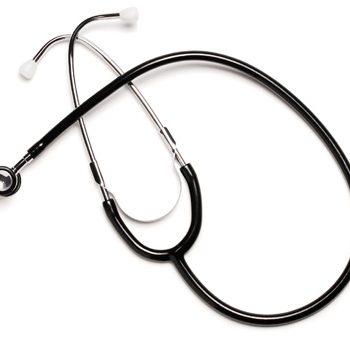 #Steth Neo-Natal Dual Head Bla Labtron
1 × $11.50
#Steth Neo-Natal Dual Head Bla Labtron
1 × $11.50 -
×
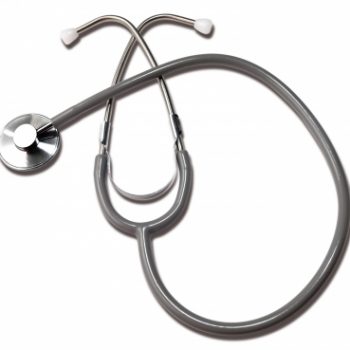 Steth Dlx Single Head Grey Labtron
1 × $5.02
Steth Dlx Single Head Grey Labtron
1 × $5.02 -
×
 Liquid Coconut Premium Oil
1 × $15.99
Liquid Coconut Premium Oil
1 × $15.99 -
×
 Bed Pkg Se Innerspring Fda Hr Lumex
1 × $1,058.00
Bed Pkg Se Innerspring Fda Hr Lumex
1 × $1,058.00 -
×
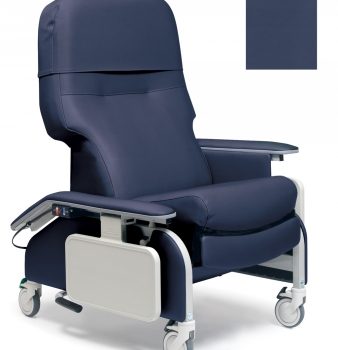 Recliner Drop Arm Imperil Blue Ca133 Lumex
1 × $2,388.00
Recliner Drop Arm Imperil Blue Ca133 Lumex
1 × $2,388.00 -
×
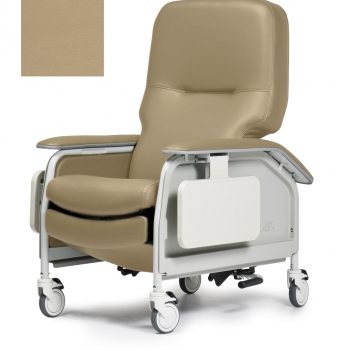 Recliner Dlx Cl Care Vntg Gold Ca133 Lumex
1 × $2,290.00
Recliner Dlx Cl Care Vntg Gold Ca133 Lumex
1 × $2,290.00 -
×
 Bed Pkg Se Rol Foam Mat Fda Hr Lumex
1 × $1,088.00
Bed Pkg Se Rol Foam Mat Fda Hr Lumex
1 × $1,088.00 -
×
 Bed Pkg Se Rol Foam Fda Fr Pl Lumex
1 × $1,130.00
Bed Pkg Se Rol Foam Fda Fr Pl Lumex
1 × $1,130.00 -
×
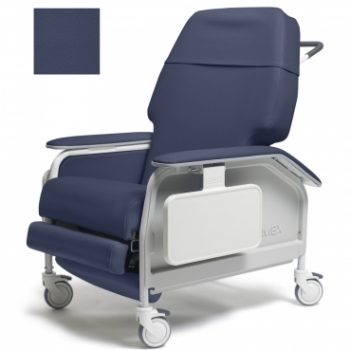 Recliner X Wd Cl Care Imp Blue Ca133 Lumex
1 × $2,422.00
Recliner X Wd Cl Care Imp Blue Ca133 Lumex
1 × $2,422.00 -
×
 Bed Pkg Se Innerspring No Rail Lumex
1 × $980.00
Bed Pkg Se Innerspring No Rail Lumex
1 × $980.00 -
×
 Rolling Utility Cart Blue Lumex
1 × $48.50
Rolling Utility Cart Blue Lumex
1 × $48.50
Subtotal: $13,631.01

