-
×
 Footrest With Composite Footplate
1 × $87.50
Footrest With Composite Footplate
1 × $87.50 -
×
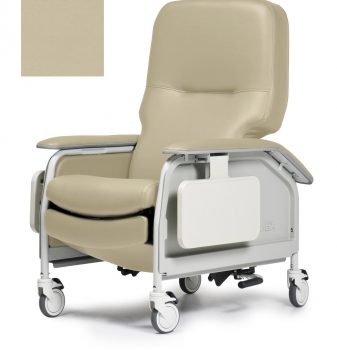 Recliner Dlx Cl Care Wm Taupe Ca133 Lumex
1 × $2,290.00
Recliner Dlx Cl Care Wm Taupe Ca133 Lumex
1 × $2,290.00 -
×
 Bed Storage Cart Lumex
1 × $68.00
Bed Storage Cart Lumex
1 × $68.00 -
×
 EfaGold (R) Flax Oil Super Lignan
1 × $22.99
EfaGold (R) Flax Oil Super Lignan
1 × $22.99 -
×
 Otoscope Set 41160N Gowllands
1 × $298.24
Otoscope Set 41160N Gowllands
1 × $298.24 -
×
 Hot Water Bottle Plybag 12Cs Grafco
1 × $83.74
Hot Water Bottle Plybag 12Cs Grafco
1 × $83.74 -
×
 Trach Jcksn Shrt Stnls Stl #5 John Bunn
1 × $136.54
Trach Jcksn Shrt Stnls Stl #5 John Bunn
1 × $136.54 -
×
 #Home Bp Kit Lot#
1 × $25.14
#Home Bp Kit Lot#
1 × $25.14 -
×
 Willow
1 × $9.99
Willow
1 × $9.99 -
×
 Vitamin D3 2,000 IU
3 × $15.49
Vitamin D3 2,000 IU
3 × $15.49 -
×
 Vitamin C-500 with Rose Hips
1 × $20.99
Vitamin C-500 with Rose Hips
1 × $20.99 -
×
 Zinc Chelate
1 × $7.49
Zinc Chelate
1 × $7.49 -
×
 Wild Yam Root
2 × $16.49
Wild Yam Root
2 × $16.49 -
×
 Zinc Lozenges
2 × $5.99
Zinc Lozenges
2 × $5.99 -
×
 Vitamin E 400 IU
1 × $14.49
Vitamin E 400 IU
1 × $14.49 -
×
 Vitamin D3, 400 IU
1 × $8.49
Vitamin D3, 400 IU
1 × $8.49 -
×
 Vitamin E 400 IU
1 × $21.99
Vitamin E 400 IU
1 × $21.99 -
×
 Wild Yam Root
1 × $9.99
Wild Yam Root
1 × $9.99
Subtotal: $3,197.01

