-
×
 Calcium Mag & D Complex
1 × $23.99
Calcium Mag & D Complex
1 × $23.99 -
×
 Shoulder Brace (L) (Size: Large)
1 × $18.20
Shoulder Brace (L) (Size: Large)
1 × $18.20 -
×
 W/C Trav Se 18X16 Fxd D-Ftr E&J
1 × $248.00
W/C Trav Se 18X16 Fxd D-Ftr E&J
1 × $248.00 -
×
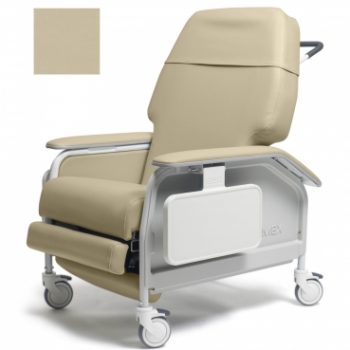 Recliner X Wd Cl Care Wm Taupe Ca133 Lumex
1 × $2,422.00
Recliner X Wd Cl Care Wm Taupe Ca133 Lumex
1 × $2,422.00 -
×
 Calcium Citrate
1 × $10.99
Calcium Citrate
1 × $10.99 -
×
 Bed Pkg Se Innerspring Fr Lumex
1 × $1,064.00
Bed Pkg Se Innerspring Fr Lumex
1 × $1,064.00 -
×
 Finger Cots Reinf Latex Lge#31 Grafco
1 × $33.56
Finger Cots Reinf Latex Lge#31 Grafco
1 × $33.56 -
×
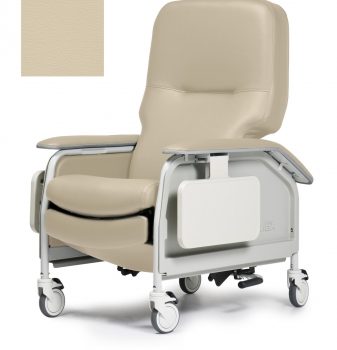 Recliner Dlx Cl Care Sand Ca133 Lumex
1 × $2,290.00
Recliner Dlx Cl Care Sand Ca133 Lumex
1 × $2,290.00 -
×
 W/C Trav L3 18X16 Full Elr E&J
1 × $326.00
W/C Trav L3 18X16 Full Elr E&J
1 × $326.00 -
×
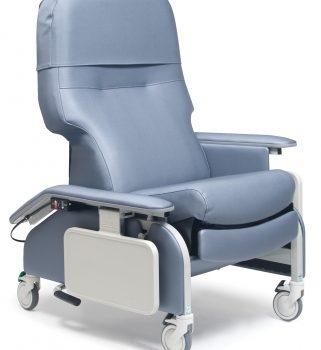 Recl Dr Arm Heat&Mas Slv Taupe Ca-133, Lumex
1 × $3,744.00
Recl Dr Arm Heat&Mas Slv Taupe Ca-133, Lumex
1 × $3,744.00 -
×
 W/C Advant Rcl 16X17 Full Elr Notify Cust Of High Freight
1 × $530.00
W/C Advant Rcl 16X17 Full Elr Notify Cust Of High Freight
1 × $530.00 -
×
 Ice Cap English Style 6" Healthteam
1 × $7.00
Ice Cap English Style 6" Healthteam
1 × $7.00 -
×
 Hot Water Bottle Boxed Grafco
1 × $7.30
Hot Water Bottle Boxed Grafco
1 × $7.30 -
×
 Bed Pkg Se Roll Foam Mat Fr Lumex
1 × $1,094.00
Bed Pkg Se Roll Foam Mat Fr Lumex
1 × $1,094.00 -
×
 Patriot Homecare Beds, Full-Electric/Low Beds
1 × $2,665.00
Patriot Homecare Beds, Full-Electric/Low Beds
1 × $2,665.00 -
×
 Bed Pkg Se Innerspring Hr Lumex
1 × $1,062.00
Bed Pkg Se Innerspring Hr Lumex
1 × $1,062.00 -
×
 Calcium Mag & Zinc
1 × $23.99
Calcium Mag & Zinc
1 × $23.99 -
×
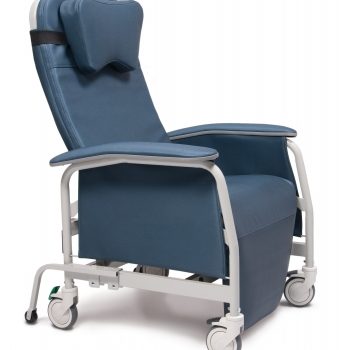 Recliner Pc Xwide Sage Ca-133, Lumex
1 × $1,870.00
Recliner Pc Xwide Sage Ca-133, Lumex
1 × $1,870.00 -
×
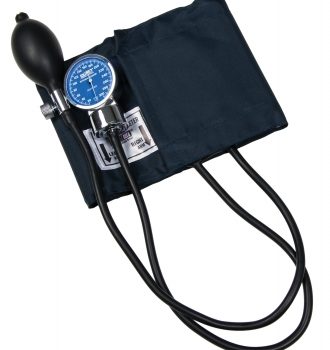 #Sphyg Optimax Chrm Gauge Bla Labtron
1 × $35.92
#Sphyg Optimax Chrm Gauge Bla Labtron
1 × $35.92 -
×
 Pat Lx Full Hc Bed Innspg Fr Lumex
1 × $1,220.00
Pat Lx Full Hc Bed Innspg Fr Lumex
1 × $1,220.00 -
×
 Recl Ortho-Bio Ii Pad Arm Euca Lumex Savoy Eucalyptus Ca133
1 × $3,522.00
Recl Ortho-Bio Ii Pad Arm Euca Lumex Savoy Eucalyptus Ca133
1 × $3,522.00 -
×
 Bed Pkg Fe Rol Fm Mt Pl Fda Fr Lumex
1 × $1,310.00
Bed Pkg Fe Rol Fm Mt Pl Fda Fr Lumex
1 × $1,310.00
Subtotal: $23,527.95

