-
×
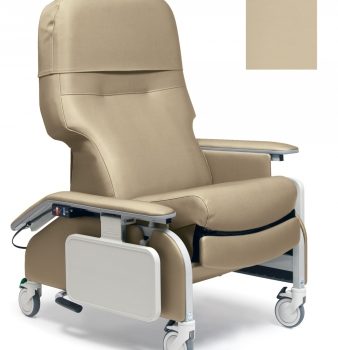 Recliner Drop Arm Warm Taupe Ca133 Lumex
1 × $2,388.00
Recliner Drop Arm Warm Taupe Ca133 Lumex
1 × $2,388.00 -
×
 Yarrow Flowers
1 × $5.99
Yarrow Flowers
1 × $5.99 -
×
 Vitamin E 400 IU
1 × $14.49
Vitamin E 400 IU
1 × $14.49 -
×
 White Kidney Bean
1 × $19.99
White Kidney Bean
1 × $19.99 -
×
 Vitamin D3, 400 IU
1 × $8.49
Vitamin D3, 400 IU
1 × $8.49 -
×
 Vitamin C 500 mg with Bioflavonoids
1 × $10.49
Vitamin C 500 mg with Bioflavonoids
1 × $10.49 -
×
 Vitex Fruit
1 × $8.49
Vitex Fruit
1 × $8.49 -
×
 Willow Standardized
1 × $23.99
Willow Standardized
1 × $23.99 -
×
 Vitamin C-500 with Rose Hips
1 × $20.99
Vitamin C-500 with Rose Hips
1 × $20.99 -
×
 Vitamin C-500 with Rose Hips
1 × $9.49
Vitamin C-500 with Rose Hips
1 × $9.49 -
×
 Zinc Lozenges
1 × $5.99
Zinc Lozenges
1 × $5.99 -
×
 Willow
1 × $9.99
Willow
1 × $9.99 -
×
 Vitamin E 400 IU
1 × $21.99
Vitamin E 400 IU
1 × $21.99 -
×
 Yucca Stalk
1 × $15.49
Yucca Stalk
1 × $15.49 -
×
 Vitamin D3 2,000 IU
1 × $15.49
Vitamin D3 2,000 IU
1 × $15.49 -
×
 White Oak Bark
1 × $9.49
White Oak Bark
1 × $9.49 -
×
 Wild Yam Root
1 × $9.99
Wild Yam Root
1 × $9.99
Subtotal: $2,598.84

