-
×
 B&T Daytime Cough & Bronchial Syrup
1 × $8.49
B&T Daytime Cough & Bronchial Syrup
1 × $8.49 -
×
 Trach Jcksn Impv Stnls Stl #5 John Bunn
2 × $118.66
Trach Jcksn Impv Stnls Stl #5 John Bunn
2 × $118.66 -
×
 Recl Ortho-Bio Ii Dolce Jet Ca-133, Lumex
2 × $3,616.00
Recl Ortho-Bio Ii Dolce Jet Ca-133, Lumex
2 × $3,616.00 -
×
 Recl Ortho-Bio Ii Blue Ridge Ca-133, Lumex
1 × $3,522.00
Recl Ortho-Bio Ii Blue Ridge Ca-133, Lumex
1 × $3,522.00 -
×
 First Aid Kitplstic-50 Person Grafco
1 × $83.04
First Aid Kitplstic-50 Person Grafco
1 × $83.04 -
×
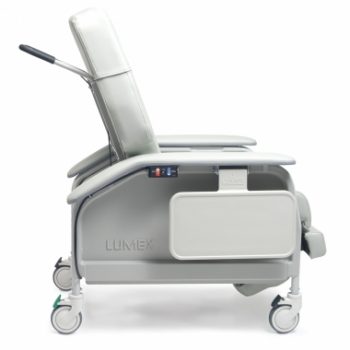 Recl Xwd Heat&Mas Warm Taupe Ca-133, Lumex
1 × $3,732.00
Recl Xwd Heat&Mas Warm Taupe Ca-133, Lumex
1 × $3,732.00 -
×
 Completia (R) Diabetic Multivitamin
1 × $29.99
Completia (R) Diabetic Multivitamin
1 × $29.99 -
×
 Echinacea Astragalus & Reishi
2 × $11.99
Echinacea Astragalus & Reishi
2 × $11.99 -
×
 Echinacea Goldenseal
1 × $18.99
Echinacea Goldenseal
1 × $18.99 -
×
 Bed Pkg Fe Innrsprng Pl Fda Hr Lumex
1 × $1,278.00
Bed Pkg Fe Innrsprng Pl Fda Hr Lumex
1 × $1,278.00 -
×
 Oxy Cylinder Carrier For W/C John Bunn
1 × $42.00
Oxy Cylinder Carrier For W/C John Bunn
1 × $42.00 -
×
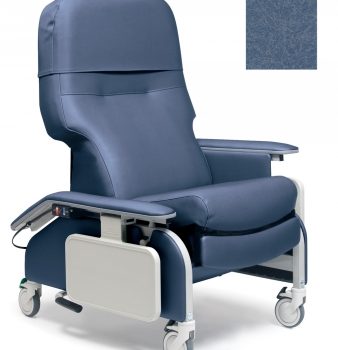 Recliner Drop Arm Steel Blue Ca133 Lumex
1 × $2,388.00
Recliner Drop Arm Steel Blue Ca133 Lumex
1 × $2,388.00 -
×
 Urinal Male Plastic W/Cvr D Grafco
1 × $3.20
Urinal Male Plastic W/Cvr D Grafco
1 × $3.20 -
×
 Traveler Recliner ®
1 × $581.00
Traveler Recliner ®
1 × $581.00 -
×
 Recliner Cl Care Terra Cotta Ca133 Lumex
1 × $2,068.00
Recliner Cl Care Terra Cotta Ca133 Lumex
1 × $2,068.00 -
×
 Pvc Hamper Std W/Lid + Bag Lumex
1 × $280.00
Pvc Hamper Std W/Lid + Bag Lumex
1 × $280.00 -
×
 Recliner Cl Care Granny Smith Ca133 Lumex
1 × $2,068.00
Recliner Cl Care Granny Smith Ca133 Lumex
1 × $2,068.00 -
×
 Trach Jcksn Impv Stnls Stl #4 John Bunn
1 × $118.66
Trach Jcksn Impv Stnls Stl #4 John Bunn
1 × $118.66 -
×
 Recliner Drop Arm Concrete Ca133 Lumex
1 × $2,388.00
Recliner Drop Arm Concrete Ca133 Lumex
1 × $2,388.00 -
×
 W/C Trans Chair Pink Alum 19 E&J
1 × $228.00
W/C Trans Chair Pink Alum 19 E&J
1 × $228.00 -
×
 Hot/Cold Neck Wrap Lumex
1 × $23.90
Hot/Cold Neck Wrap Lumex
1 × $23.90 -
×
 Thera-Putty 2 Oz Med-Firm Grn Lumex
1 × $6.56
Thera-Putty 2 Oz Med-Firm Grn Lumex
1 × $6.56 -
×
 Lumbar Cushion (Foam) 16X17 Lumex
1 × $52.00
Lumbar Cushion (Foam) 16X17 Lumex
1 × $52.00 -
×
 W/C Skin Gel Pro Cushion E & J Duragel Spiii
1 × $120.00
W/C Skin Gel Pro Cushion E & J Duragel Spiii
1 × $120.00 -
×
 Vitamin D3 5,000 IU
1 × $18.99
Vitamin D3 5,000 IU
1 × $18.99 -
×
 W/C Trans Bariatric Chair 24W Al E&J **On Temporary Hold**
1 × $440.00
W/C Trans Bariatric Chair 24W Al E&J **On Temporary Hold**
1 × $440.00
Subtotal: $26,992.12