-
×
 #Lg Adult Cuff For 1103
1 × $18.12
#Lg Adult Cuff For 1103
1 × $18.12 -
×
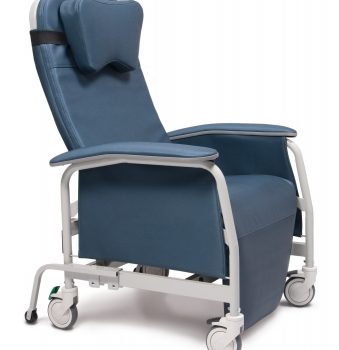 Recliner Pc Xwide Meteor Ca-133, Lumex
1 × $1,870.00
Recliner Pc Xwide Meteor Ca-133, Lumex
1 × $1,870.00 -
×
 Recl Ortho-Bio Ii Fence Ca-133, Lumex
1 × $3,616.00
Recl Ortho-Bio Ii Fence Ca-133, Lumex
1 × $3,616.00 -
×
 Primadophilus (R) Kids Cherry Chew
1 × $9.99
Primadophilus (R) Kids Cherry Chew
1 × $9.99 -
×
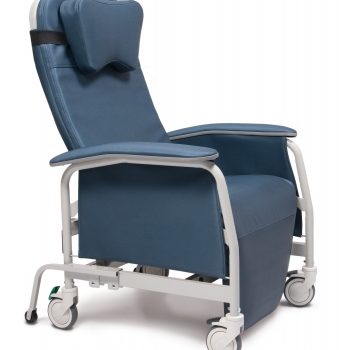 Recliner Pc Xwide Periwinkle Ca-133, Lumex
1 × $1,776.00
Recliner Pc Xwide Periwinkle Ca-133, Lumex
1 × $1,776.00 -
×
 Recl Heat&Mas Vintage Gold Ca-133, Lumex
1 × $3,318.00
Recl Heat&Mas Vintage Gold Ca-133, Lumex
1 × $3,318.00 -
×
 Alpha (R) CF
1 × $11.49
Alpha (R) CF
1 × $11.49 -
×
 Bed Pkg Fe Innrsprng Pl Fda Fr Lumex
1 × $1,280.00
Bed Pkg Fe Innrsprng Pl Fda Fr Lumex
1 × $1,280.00 -
×
 Alive! (R) Whey Protein (Berry)
1 × $37.99
Alive! (R) Whey Protein (Berry)
1 × $37.99 -
×
 Ginkgold (R)
1 × $40.49
Ginkgold (R)
1 × $40.49 -
×
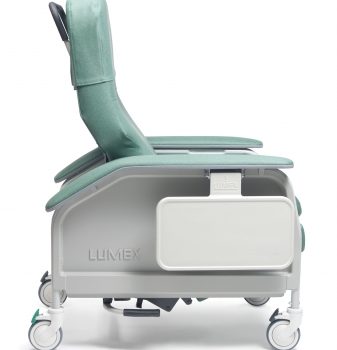 Recl Dlx Heat&Mas Gypsum Ca-133, Lumex
1 × $3,474.00
Recl Dlx Heat&Mas Gypsum Ca-133, Lumex
1 × $3,474.00 -
×
 W/C Navigator 18W Blue E&J
1 × $490.00
W/C Navigator 18W Blue E&J
1 × $490.00 -
×
 Elevating Legrest With Composite Footplate
1 × $137.50
Elevating Legrest With Composite Footplate
1 × $137.50 -
×
 W/C Rsc Ss Hi-Bk Pin Lk E&J
1 × $1,382.00
W/C Rsc Ss Hi-Bk Pin Lk E&J
1 × $1,382.00 -
×
 Trach Jcksn Impv Stnls Stl #5 John Bunn
1 × $118.66
Trach Jcksn Impv Stnls Stl #5 John Bunn
1 × $118.66 -
×
 W/C Rsc Ss Hi-Bk E&J
1 × $1,328.00
W/C Rsc Ss Hi-Bk E&J
1 × $1,328.00 -
×
 Trach Jcksn Impv Stnls Stl #4 John Bunn
1 × $118.66
Trach Jcksn Impv Stnls Stl #4 John Bunn
1 × $118.66 -
×
 Trach Jcksn Impv Stnls Stl #6 John Bunn
1 × $118.66
Trach Jcksn Impv Stnls Stl #6 John Bunn
1 × $118.66 -
×
 W/C Traveler Se 18X16 Fxd Fxd E&J
1 × $244.00
W/C Traveler Se 18X16 Fxd Fxd E&J
1 × $244.00 -
×
 Traveler ®
1 × $471.00
Traveler ®
1 × $471.00 -
×
 Neb-U-Tyke Ic - Penguin Pediatric Nebulizer
1 × $57.00
Neb-U-Tyke Ic - Penguin Pediatric Nebulizer
1 × $57.00 -
×
 Neb-U-Tyke Bella Butterfly John Bunn
1 × $61.90
Neb-U-Tyke Bella Butterfly John Bunn
1 × $61.90 -
×
 Regulator H Cga540 0-8 W/Gauge John Bunn
1 × $50.00
Regulator H Cga540 0-8 W/Gauge John Bunn
1 × $50.00 -
×
 Vitamin B2
1 × $8.49
Vitamin B2
1 × $8.49 -
×
 Digi02 Finger Pulse Oximeter John Bunn
1 × $292.50
Digi02 Finger Pulse Oximeter John Bunn
1 × $292.50 -
×
 Vitamin C-500 with Rose Hips
1 × $20.99
Vitamin C-500 with Rose Hips
1 × $20.99 -
×
 Willow
2 × $9.99
Willow
2 × $9.99 -
×
 Vitamin D3 2,000 IU
1 × $8.99
Vitamin D3 2,000 IU
1 × $8.99
Subtotal: $20,380.41

