-
×
 Recl Xwd Heat&Mas Royal Blue Ca-133, Lumex
1 × $3,732.00
Recl Xwd Heat&Mas Royal Blue Ca-133, Lumex
1 × $3,732.00 -
×
 Recliner Dlx Cl Care Port Ca133 Lumex
1 × $2,290.00
Recliner Dlx Cl Care Port Ca133 Lumex
1 × $2,290.00 -
×
 Recl Ortho-Bio Ii Pad Arm Jet Lumex Dolce Meteor Ca133
1 × $3,616.00
Recl Ortho-Bio Ii Pad Arm Jet Lumex Dolce Meteor Ca133
1 × $3,616.00 -
×
 Recliner Cl Care Warm Taupe Ca133 New Ver Lumex
1 × $2,068.00
Recliner Cl Care Warm Taupe Ca133 New Ver Lumex
1 × $2,068.00 -
×
 W/C Trans Black Alum 19 E&J
1 × $228.00
W/C Trans Black Alum 19 E&J
1 × $228.00 -
×
 Recl Dlx Heat&Mas Eucalyptus Ca-133, Lumex
1 × $3,474.00
Recl Dlx Heat&Mas Eucalyptus Ca-133, Lumex
1 × $3,474.00 -
×
 Recliner Cl Care Concrete Ca133 Lumex
1 × $2,068.00
Recliner Cl Care Concrete Ca133 Lumex
1 × $2,068.00 -
×
 Bed Pkg Bariatric Lumex
1 × $3,650.00
Bed Pkg Bariatric Lumex
1 × $3,650.00 -
×
 Positioning Belt (Auto Style)
1 × $26.00
Positioning Belt (Auto Style)
1 × $26.00 -
×
 Recl X Wd Cl Care Sea Ca-133, Lumex
1 × $2,422.00
Recl X Wd Cl Care Sea Ca-133, Lumex
1 × $2,422.00 -
×
 Recl Heat&Mas Port Ca-133, Lumex
1 × $3,318.00
Recl Heat&Mas Port Ca-133, Lumex
1 × $3,318.00 -
×
 Mirror Regal 3-In-1 Grafco
1 × $27.12
Mirror Regal 3-In-1 Grafco
1 × $27.12 -
×
 Pat Lx Full Hc Bed Rol Foam Hr Lumex
1 × $1,276.00
Pat Lx Full Hc Bed Rol Foam Hr Lumex
1 × $1,276.00 -
×
 EfaGold (R) Krill Oil
1 × $41.49
EfaGold (R) Krill Oil
1 × $41.49 -
×
 Recliner Pc Xwide Steel Grey Ca-133, Lumex
1 × $1,776.00
Recliner Pc Xwide Steel Grey Ca-133, Lumex
1 × $1,776.00 -
×
 Recliner Cl Care Terra Cotta Ca133 Lumex
1 × $2,068.00
Recliner Cl Care Terra Cotta Ca133 Lumex
1 × $2,068.00 -
×
 Recliner Pc Xwide Warm Taupe Ca-133, Lumex
1 × $1,776.00
Recliner Pc Xwide Warm Taupe Ca-133, Lumex
1 × $1,776.00 -
×
 Recl Ortho-Bio Ii Pad Arm Peri Lumex Ren Periwinkle Ca133
1 × $3,522.00
Recl Ortho-Bio Ii Pad Arm Peri Lumex Ren Periwinkle Ca133
1 × $3,522.00 -
×
 Recl Ortho-Bio Ii Pad Arm Wine Lumex Ren Wineberry Ca133
1 × $3,522.00
Recl Ortho-Bio Ii Pad Arm Wine Lumex Ren Wineberry Ca133
1 × $3,522.00 -
×
 Recl X Wd Cl Care Sand Ca-133, Lumex
1 × $2,422.00
Recl X Wd Cl Care Sand Ca-133, Lumex
1 × $2,422.00 -
×
 Recl Ortho-Bio Ii Pad Arm Armo Lumex Guardian Convoy Ca133
1 × $3,616.00
Recl Ortho-Bio Ii Pad Arm Armo Lumex Guardian Convoy Ca133
1 × $3,616.00 -
×
 Recl Ortho-Bio Ii Mulberry Ca-133, Lumex
1 × $3,522.00
Recl Ortho-Bio Ii Mulberry Ca-133, Lumex
1 × $3,522.00 -
×
 Curica (R) Pain Relief
1 × $42.99
Curica (R) Pain Relief
1 × $42.99 -
×
 Recl X Wd Cl Care Champagne Ca-133, Lumex
1 × $2,516.00
Recl X Wd Cl Care Champagne Ca-133, Lumex
1 × $2,516.00 -
×
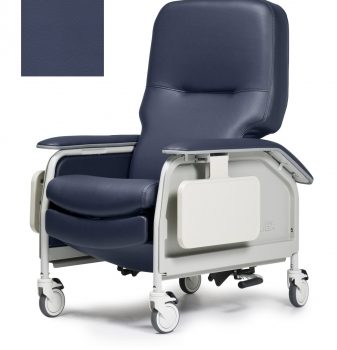 Recliner Dlx Cl Care Impl Blue Ca133 Lumex
1 × $2,290.00
Recliner Dlx Cl Care Impl Blue Ca133 Lumex
1 × $2,290.00 -
×
 Shoulder Brace
1 × $18.20
Shoulder Brace
1 × $18.20 -
×
 Black Cohosh Root
1 × $15.49
Black Cohosh Root
1 × $15.49 -
×
 Bathtub Rail Sure Safe Lumex
1 × $45.70
Bathtub Rail Sure Safe Lumex
1 × $45.70 -
×
 SssstingStop (R)
1 × $7.49
SssstingStop (R)
1 × $7.49 -
×
 #Sphyg Blk Nyl Lg Ad W/Gauge G Latex Free, Labtron
1 × $33.42
#Sphyg Blk Nyl Lg Ad W/Gauge G Latex Free, Labtron
1 × $33.42 -
×
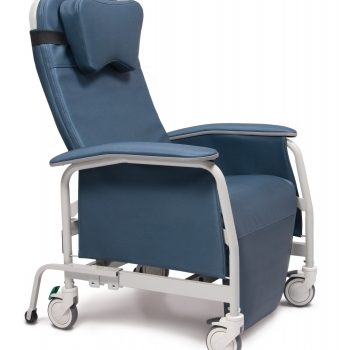 Recliner Pc Xwide Steel Blue Ca-133, Lumex
1 × $1,776.00
Recliner Pc Xwide Steel Blue Ca-133, Lumex
1 × $1,776.00 -
×
 Recl Ortho-Bio Ii Pad Arm Cypr Lumex Ren Cypress Ca133
1 × $3,522.00
Recl Ortho-Bio Ii Pad Arm Cypr Lumex Ren Cypress Ca133
1 × $3,522.00 -
×
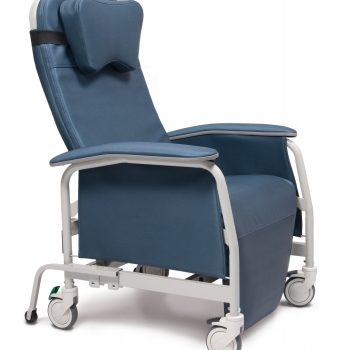 Recliner Pc Xwide Impl Blue Ca-133, Lumex
1 × $1,776.00
Recliner Pc Xwide Impl Blue Ca-133, Lumex
1 × $1,776.00
Subtotal: $62,503.90

