-
×
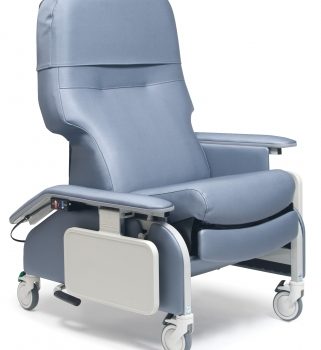 Recl Dr Arm Heat&Mas Grn Smith Ca-133, Lumex
1 × $3,648.00
Recl Dr Arm Heat&Mas Grn Smith Ca-133, Lumex
1 × $3,648.00 -
×
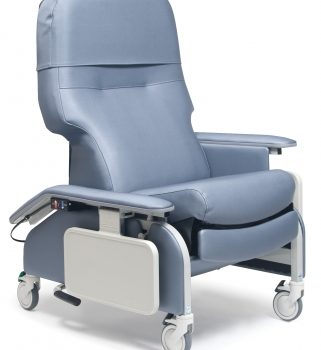 Recl Dr Arm Heat&Mas Sea Ca-133, Lumex
1 × $3,648.00
Recl Dr Arm Heat&Mas Sea Ca-133, Lumex
1 × $3,648.00 -
×
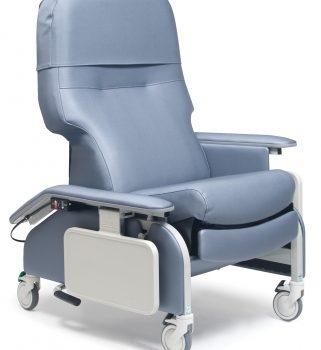 Recl Dr Arm Heat&Mas Slv Taupe Ca-133, Lumex
1 × $3,744.00
Recl Dr Arm Heat&Mas Slv Taupe Ca-133, Lumex
1 × $3,744.00 -
×
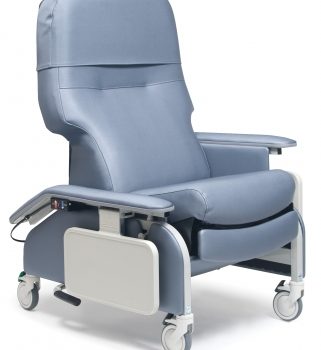 Recl Dr Arm Heat&Mas Slv Jet Ca-133, Lumex
1 × $3,744.00
Recl Dr Arm Heat&Mas Slv Jet Ca-133, Lumex
1 × $3,744.00 -
×
 Mirror Regal 3-In-1 Grafco
1 × $27.12
Mirror Regal 3-In-1 Grafco
1 × $27.12 -
×
 Cooling Pad Lumex
1 × $23.90
Cooling Pad Lumex
1 × $23.90 -
×
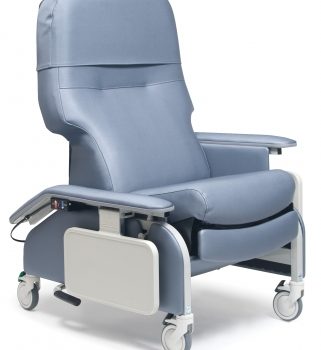 Recl Dr Arm Heat&Mas Wineberry Ca133 Lumex
1 × $3,648.00
Recl Dr Arm Heat&Mas Wineberry Ca133 Lumex
1 × $3,648.00 -
×
 Thera-Putty 4 Oz Med-Firm Grn Lumex
1 × $9.30
Thera-Putty 4 Oz Med-Firm Grn Lumex
1 × $9.30 -
×
 Thera-Putty 4 Oz X-Soft Beige Lumex
1 × $9.30
Thera-Putty 4 Oz X-Soft Beige Lumex
1 × $9.30 -
×
 Cover Terry Cloth Grafco
1 × $32.00
Cover Terry Cloth Grafco
1 × $32.00 -
×
 Vitamin E 400 IU
1 × $21.99
Vitamin E 400 IU
1 × $21.99 -
×
 Vitamin D3 2,000 IU
1 × $15.49
Vitamin D3 2,000 IU
1 × $15.49 -
×
 Willow
1 × $9.99
Willow
1 × $9.99
Subtotal: $18,581.09

