-
×
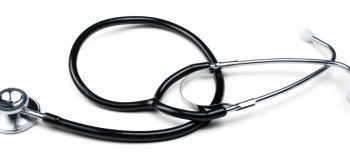 #Dualhead Steth Black Lot#
1 × $7.60
#Dualhead Steth Black Lot#
1 × $7.60 -
×
 Trach Jcksn Shrt Stnls Stl #8 John Bunn
1 × $136.54
Trach Jcksn Shrt Stnls Stl #8 John Bunn
1 × $136.54 -
×
 Acetyl L-Carnitine
1 × $25.99
Acetyl L-Carnitine
1 × $25.99 -
×
 5-HTP
1 × $9.99
5-HTP
1 × $9.99 -
×
 Trach Jcksn Shrt Stnls Stl #4 John Bunn
1 × $136.54
Trach Jcksn Shrt Stnls Stl #4 John Bunn
1 × $136.54 -
×
 Black Sprague Stethoscope Lot
1 × $14.16
Black Sprague Stethoscope Lot
1 × $14.16 -
×
 W/C Trans Chair Red Alum 17 E&J
1 × $228.00
W/C Trans Chair Red Alum 17 E&J
1 × $228.00 -
×
 Portable Ultrasonic Nebulizer
1 × $24.95
Portable Ultrasonic Nebulizer
1 × $24.95 -
×
 Digital Thermometer/Quick Read Lot #
1 × $8.18
Digital Thermometer/Quick Read Lot #
1 × $8.18 -
×
 #Aneroid Sphyg W/Inf Cuff Lot#
1 × $22.38
#Aneroid Sphyg W/Inf Cuff Lot#
1 × $22.38 -
×
 W/C Trans Silver Vein Steel 17 E&J
2 × $184.00
W/C Trans Silver Vein Steel 17 E&J
2 × $184.00 -
×
 Digi02 Finger Pulse Oximeter John Bunn
2 × $292.50
Digi02 Finger Pulse Oximeter John Bunn
2 × $292.50 -
×
 Thera-Putty 4 Oz Soft-Med Red Lumex
1 × $9.30
Thera-Putty 4 Oz Soft-Med Red Lumex
1 × $9.30 -
×
 Rollator Alum Wide Black Walkabout Wide
1 × $146.00
Rollator Alum Wide Black Walkabout Wide
1 × $146.00 -
×
 Yarrow Flowers
2 × $5.99
Yarrow Flowers
2 × $5.99 -
×
 Yucca Stalk
1 × $10.49
Yucca Stalk
1 × $10.49 -
×
 Lumbar Cushion (Foam) 16X17 Lumex
1 × $52.00
Lumbar Cushion (Foam) 16X17 Lumex
1 × $52.00 -
×
 Elevating Legrest With Composite Footplate
1 × $137.50
Elevating Legrest With Composite Footplate
1 × $137.50 -
×
 W/C Trans Chair Silvr Alum 17 E&J
1 × $228.00
W/C Trans Chair Silvr Alum 17 E&J
1 × $228.00 -
×
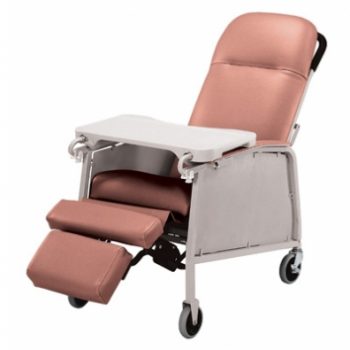 Recliner Std 3 Pos Rosewood Lumex
1 × $638.00
Recliner Std 3 Pos Rosewood Lumex
1 × $638.00 -
×
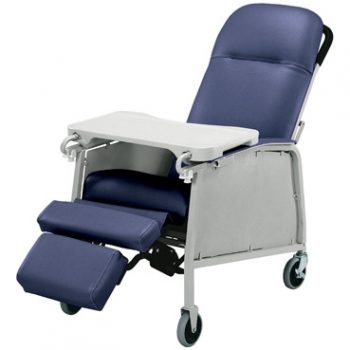 Recliner Std 3 Pos Imperl Blue Lumex
1 × $638.00
Recliner Std 3 Pos Imperl Blue Lumex
1 × $638.00 -
×
 Vitamin D3, 400 IU
1 × $8.49
Vitamin D3, 400 IU
1 × $8.49 -
×
 Willow Standardized
1 × $23.99
Willow Standardized
1 × $23.99
Subtotal: $3,471.08

