-
×
 Alive! (R) Ultra-Shake (TM) Pea Protein
1 × $25.99
Alive! (R) Ultra-Shake (TM) Pea Protein
1 × $25.99 -
×
 Recl Xwd Heat&Mas Sand Ca-133, Lumex
1 × $3,732.00
Recl Xwd Heat&Mas Sand Ca-133, Lumex
1 × $3,732.00 -
×
 Steth Stainless Steel Adult Labtron
1 × $40.94
Steth Stainless Steel Adult Labtron
1 × $40.94 -
×
 #Steth Disposable Champ Blk Labtron
1 × $4.40
#Steth Disposable Champ Blk Labtron
1 × $4.40 -
×
 Recl Xwd Heat&Mas Gypsum Ca-133, Lumex
1 × $3,732.00
Recl Xwd Heat&Mas Gypsum Ca-133, Lumex
1 × $3,732.00 -
×
 Recliner X Wd Cl Care Bl Rdg Ca133 Lumex
1 × $2,422.00
Recliner X Wd Cl Care Bl Rdg Ca133 Lumex
1 × $2,422.00 -
×
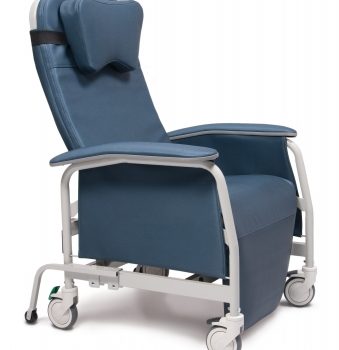 Recliner Pc Xwide Warm Taupe Ca-133, Lumex
1 × $1,776.00
Recliner Pc Xwide Warm Taupe Ca-133, Lumex
1 × $1,776.00 -
×
 Scissor Nail Grafco
1 × $7.24
Scissor Nail Grafco
1 × $7.24 -
×
 Comfort Cushion 20X16X3 E&J
1 × $34.00
Comfort Cushion 20X16X3 E&J
1 × $34.00 -
×
 Recliner Cl Care Slv Sage Ca133 Lumex
1 × $2,118.00
Recliner Cl Care Slv Sage Ca133 Lumex
1 × $2,118.00 -
×
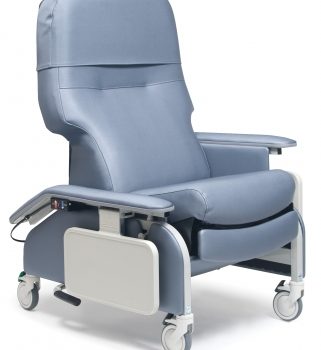 Recl Dr Arm Heat&Mas Slv Jet Ca-133, Lumex
1 × $3,744.00
Recl Dr Arm Heat&Mas Slv Jet Ca-133, Lumex
1 × $3,744.00 -
×
 Echinacea Purpurea Herb
1 × $12.49
Echinacea Purpurea Herb
1 × $12.49 -
×
 Broad Spectrum Enzyme
1 × $16.99
Broad Spectrum Enzyme
1 × $16.99 -
×
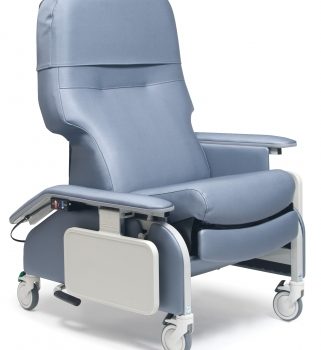 Recl Dr Arm Heat&Mas Tra Cotta Ca-133, Lumex
1 × $3,648.00
Recl Dr Arm Heat&Mas Tra Cotta Ca-133, Lumex
1 × $3,648.00 -
×
 Blood Sugar
1 × $15.99
Blood Sugar
1 × $15.99 -
×
 Recl Heat&Mas Oatmeal Ca-133, Lumex
1 × $3,318.00
Recl Heat&Mas Oatmeal Ca-133, Lumex
1 × $3,318.00 -
×
 Bed Pressure Pad For P Alarm Lumex
1 × $67.00
Bed Pressure Pad For P Alarm Lumex
1 × $67.00 -
×
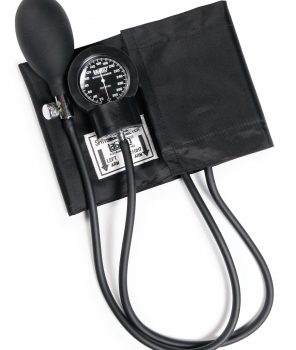 #Sphyg Blk Nyl Ad W/Gauge Guar Latex Free, Labtron
1 × $37.18
#Sphyg Blk Nyl Ad W/Gauge Guar Latex Free, Labtron
1 × $37.18 -
×
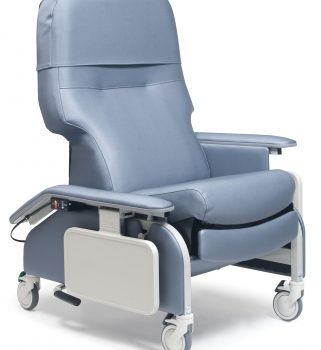 Lumex Deluxe Clinical Care Recliner With Drop Arms
1 × $2,935.00
Lumex Deluxe Clinical Care Recliner With Drop Arms
1 × $2,935.00 -
×
 Blessed Thistle
1 × $10.49
Blessed Thistle
1 × $10.49 -
×
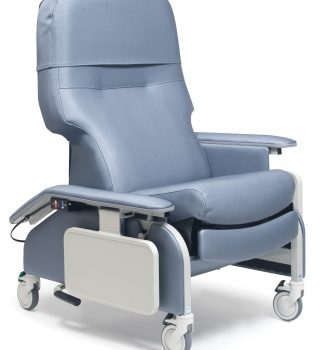 Recl Dr Arm Heat&Mas Berry Ca-133, Lumex
1 × $3,648.00
Recl Dr Arm Heat&Mas Berry Ca-133, Lumex
1 × $3,648.00 -
×
 W/C Cushion Gel/Foam18X18X4.5 E & J Duragel Spp
1 × $134.00
W/C Cushion Gel/Foam18X18X4.5 E & J Duragel Spp
1 × $134.00 -
×
 W/C Trans Silver Vein Steel 17 E&J
1 × $184.00
W/C Trans Silver Vein Steel 17 E&J
1 × $184.00 -
×
 W/C Trans Black Alum 17 E&J
1 × $228.00
W/C Trans Black Alum 17 E&J
1 × $228.00 -
×
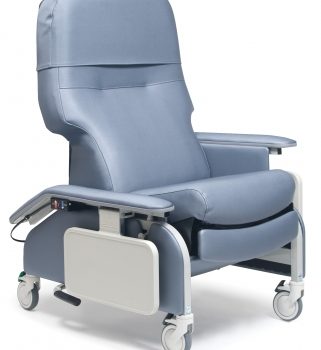 Recl Dr Arm Heat&Mas Taupe Ca-133, Lumex
1 × $3,648.00
Recl Dr Arm Heat&Mas Taupe Ca-133, Lumex
1 × $3,648.00 -
×
 Body Essential (R) Silica Caps with Calcium
1 × $20.49
Body Essential (R) Silica Caps with Calcium
1 × $20.49 -
×
 AloeMaxLax (TM) with Cascara Sagrada
1 × $12.99
AloeMaxLax (TM) with Cascara Sagrada
1 × $12.99 -
×
 W/C Trans Chair Red Alum 17 E&J
1 × $228.00
W/C Trans Chair Red Alum 17 E&J
1 × $228.00 -
×
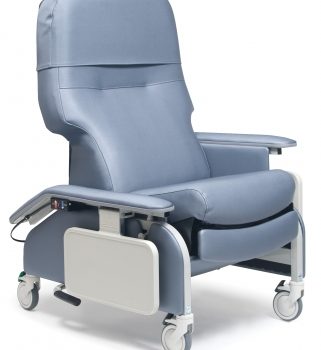 Recl Dr Arm Heat&Mas Chestnut Ca-133, Lumex
1 × $3,648.00
Recl Dr Arm Heat&Mas Chestnut Ca-133, Lumex
1 × $3,648.00 -
×
 W/C Skin Gel Pro Cushion E & J Duragel Spiii
1 × $136.00
W/C Skin Gel Pro Cushion E & J Duragel Spiii
1 × $136.00 -
×
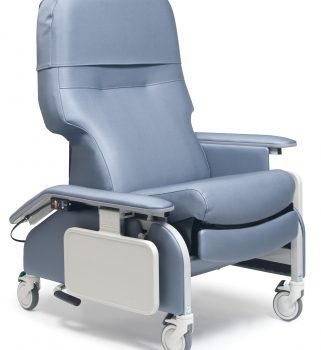 Recl Dr Arm Heat&Mas Vin Gold Ca-133, Lumex
1 × $3,648.00
Recl Dr Arm Heat&Mas Vin Gold Ca-133, Lumex
1 × $3,648.00 -
×
 Wc Skin Gel Pro Cushion E & J Duragel Spiii
1 × $128.00
Wc Skin Gel Pro Cushion E & J Duragel Spiii
1 × $128.00 -
×
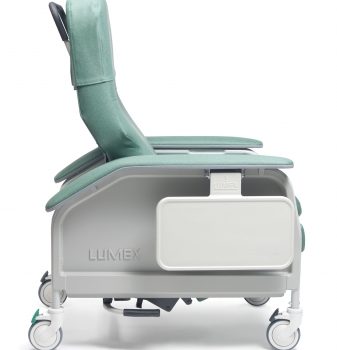 Recl Dlx Heat&Mas Cypress Ca-133, Lumex
1 × $3,474.00
Recl Dlx Heat&Mas Cypress Ca-133, Lumex
1 × $3,474.00 -
×
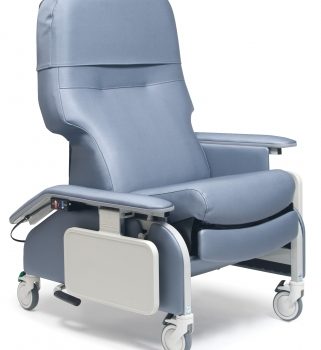 Recl Dr Arm Heat&Mas Grn Smith Ca-133, Lumex
1 × $3,648.00
Recl Dr Arm Heat&Mas Grn Smith Ca-133, Lumex
1 × $3,648.00 -
×
 W/C Skin Gel Pro Cushion E & J Duragel Spiii
1 × $136.00
W/C Skin Gel Pro Cushion E & J Duragel Spiii
1 × $136.00 -
×
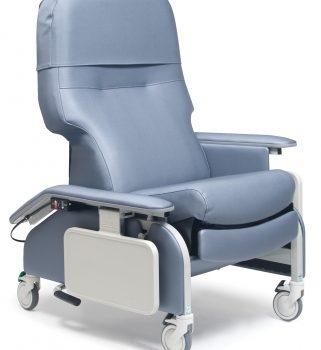 Recl Dr Arm Heat&Mas Slv Taupe Ca-133, Lumex
1 × $3,744.00
Recl Dr Arm Heat&Mas Slv Taupe Ca-133, Lumex
1 × $3,744.00 -
×
 Recl Heat&Mas Dolce Jet Ca-133, Lumex
1 × $3,130.00
Recl Heat&Mas Dolce Jet Ca-133, Lumex
1 × $3,130.00 -
×
 W/C Cushion Gel/Foam 20X16X4.5 E & J Duragel Spp
1 × $134.00
W/C Cushion Gel/Foam 20X16X4.5 E & J Duragel Spp
1 × $134.00 -
×
 W/C Skin Gel Pro Cushion E & J Duragel Spiii
1 × $124.00
W/C Skin Gel Pro Cushion E & J Duragel Spiii
1 × $124.00 -
×
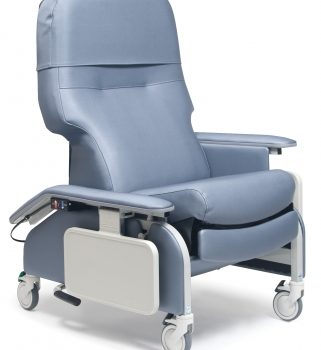 Recl Dr Arm Heat&Mas Sea Ca-133, Lumex
1 × $3,648.00
Recl Dr Arm Heat&Mas Sea Ca-133, Lumex
1 × $3,648.00 -
×
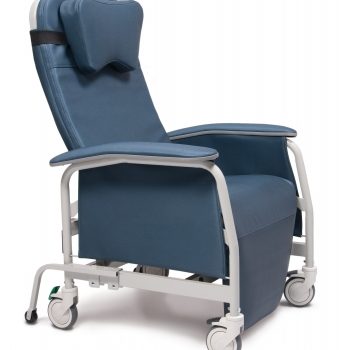 Recliner Pc Xwide Vintage Gold Ca-133, Lumex
1 × $1,776.00
Recliner Pc Xwide Vintage Gold Ca-133, Lumex
1 × $1,776.00 -
×
 Echinacea Goldenseal
1 × $8.49
Echinacea Goldenseal
1 × $8.49 -
×
 Vitamin D3, 400 IU
1 × $8.49
Vitamin D3, 400 IU
1 × $8.49 -
×
 Vitamin C 500 mg with Bioflavonoids
1 × $10.49
Vitamin C 500 mg with Bioflavonoids
1 × $10.49 -
×
 Zinc Chelate
1 × $7.49
Zinc Chelate
1 × $7.49 -
×
 Yucca Stalk
1 × $10.49
Yucca Stalk
1 × $10.49 -
×
 Vitamin B2
1 × $8.49
Vitamin B2
1 × $8.49 -
×
 Yucca Stalk
1 × $15.49
Yucca Stalk
1 × $15.49 -
×
 Vitamin C 500 mg with Bioflavonoids
1 × $22.99
Vitamin C 500 mg with Bioflavonoids
1 × $22.99 -
×
 Willow Standardized
1 × $23.99
Willow Standardized
1 × $23.99 -
×
 White Oak Bark
1 × $9.49
White Oak Bark
1 × $9.49
Subtotal: $63,301.09

